
^ Tritium occurs naturally as a cosmogenic nuclide.^ Produced during Big Bang nucleosynthesis, but not primordial, as all such atoms have since decayed to 3.^ Produced during Big Bang nucleosynthesis.^ # – Values marked # are not purely derived from experimental data, but at least partly from trends of neighboring nuclides (TNN).Īre the only stable nuclides with more protons than neutrons.^ ( ) spin value – Indicates spin with weak assignment arguments.^ Bold symbol as daughter – Daughter product is stable.^ ( ) – Uncertainty (1 σ) is given in concise form in parentheses after the corresponding last digits.(During the early study of radioactivity, some other heavy radioactive isotopes were given names, but such names are rarely used today.) 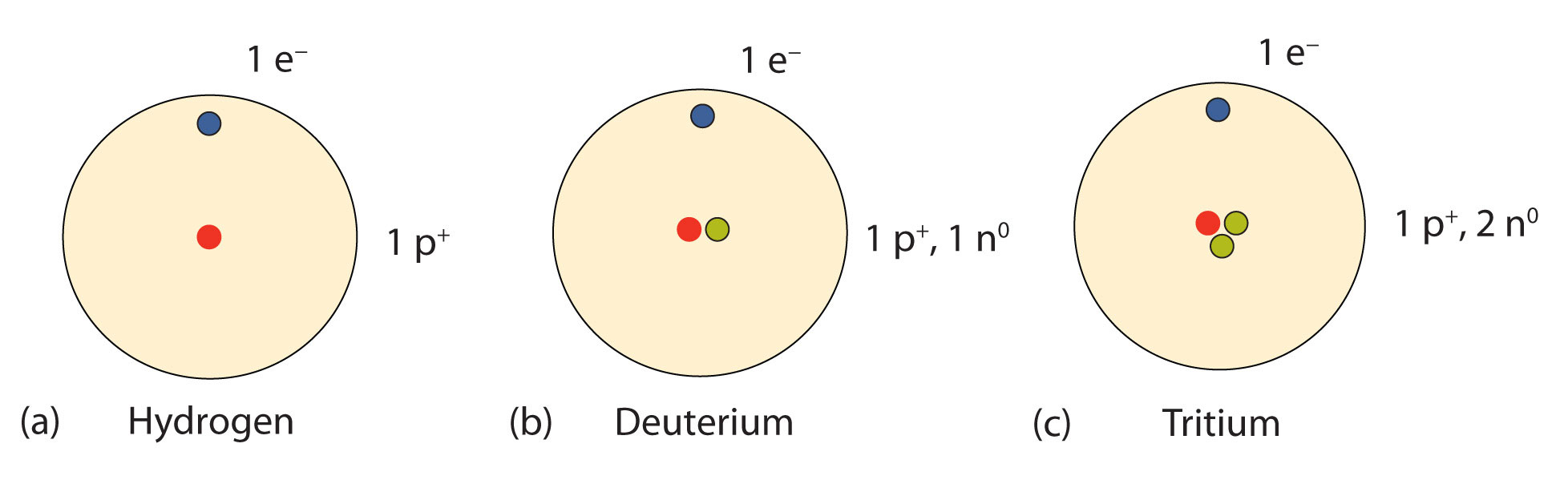
, with no neutrons, is sometimes called protium. ) instead to avoid confusion in the alphabetic sorting of chemical formulas. The IUPAC accepts the D and T symbols, but recommends using standard isotopic symbols ( 2 The symbols D and T are sometimes used for deuterium and tritium. (or hydrogen-2) isotope is deuterium and the 3 Hydrogen is the only element whose isotopes have different names that remain in common use today: the 2 Heavier isotopes also exist, all of which are synthetic and have a half-life of less than one zeptosecond (10 −21 s). Hydrogen ( 1H) has three naturally occurring isotopes, sometimes denoted 1 Consider the following relationship.The three most stable isotopes of hydrogen: protium ( A = 1), deuterium ( A = 2), and tritium ( A = 3). Therefore, the superscript can be said to represent the number of nucleons in the atom.ĭetermining the number of neutrons in an atom is a simple matter. A nucleon is a particle that resides in the nucleus, either a proton or neutron. Since the superscript is the number of protons and neutrons added together, it is convenient to have a term that describes both types of subatomic particles. Here are the symbols of the three isotopes of hydrogen. A superscript is used for the number of protons and neutrons (the mass number), and a subscript is used for the number of protons (the atomic number). There is a way to write an element symbol that indicates the number of protons and neutrons. The percent abundances of each isotope, respectively, are 25.654%, 39.156%, and 35.190%. There are three iosotopes, each with masses 65, 67, and 69. Sample ProblemĬalculate the weight averaged atomic mass of hypothetical element X. 
This calculation can be done for every element one just needs to know the masses of the isotopes and the percent abundances. Obviously, the mass of the heavier isotope contributes more to the atomic mass than the lighter isotope. This mass is comparable to the atomic mass on the periodic table. Hydrogen will be focused on for much of this introduction.Ī hydrogen atom contains one proton and one electron. The lightest element on the periodic table is hydrogen it only contains one proton. The elements on the periodic table are organized according to their atomic numbers. The mass number is the number of protons plus the number of neutrons. The atomic number is what is used to identify the element. The atomic number is the number of protons in an element. This brings us to two common terms used in chemistry, atomic number and mass number. If you know the number of protons, neutrons, and electrons in an element, it is a simple matter to calculate the mass of that element. Obviously, the mass of a proton and neutron are very similar, while the mass of an electron is much less than the other two particles. Mass of proton = 1.007277 amu = 1.6726*10 -27 kgĪmu stands for atomic mass unit.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
